

On a rocky archipelago in the North Atlantic Ocean, staff at the Faroese Environment Agency and the Faroe Marine Research Institute regularly sample tissues from the North Atlantic long-finned pilot whales that roam the waters around the islands. The archive of these samples stretches back to the 1980s and has helped researchers determine the reach of human-made contaminants in the remote marine environment.
Jennifer Sun is one of those researchers. Sun studies PFAS—per- and polyfluoroalkyl substances, commonly known as “forever chemicals”—at Harvard University and is the lead author of a recently published study that analyzed how these toxic chemicals have accumulated in pilot whale tissue over the past 2 decades.

Using samples of whale tissue collected between 2001 and 2023, Sun and her colleagues measured a parameter called bulk extractable organofluorine, which shows the overall amount of organofluorine-containing chemicals (including PFAS) in the tissue. They then used a more targeted analysis able to confirm the identity of 28 specific chemicals out of thousands of possible PFAS formulations.
The study’s results showed an expected decrease in the concentrations of older PFAS but an unexpected absence of newer PFAS chemicals. This anomaly could be indicative of an emerging question in PFAS research: Where are the newest PFAS going?
Prolific PFAS
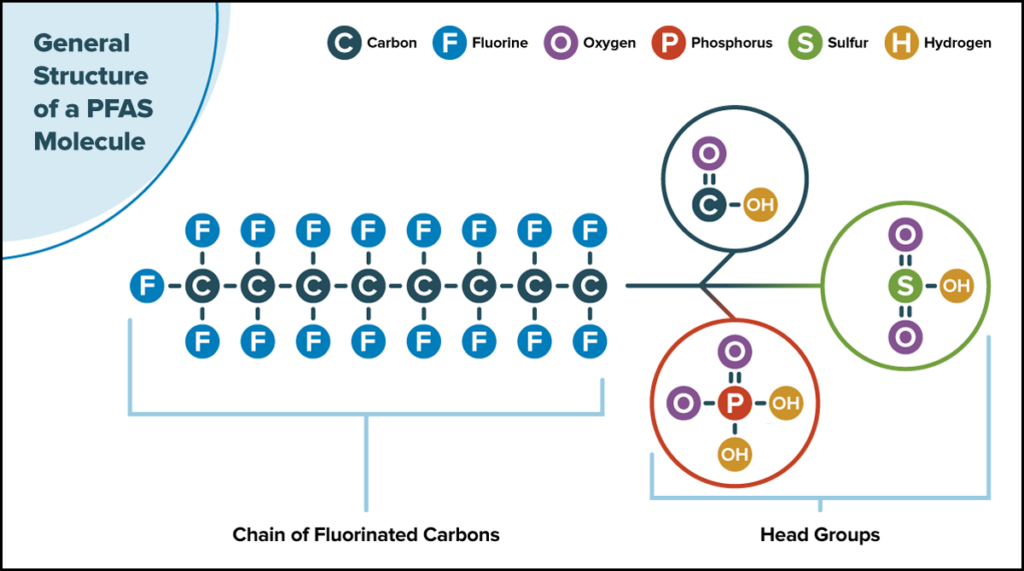
There are two general categories of PFAS. The first includes legacy PFAS such as perfluorooctanoic acid (PFOA) and perfluorooctane sulfonic acid (PFOS). Chemical manufacturers produced these compounds in the 1970s, 1980s, and 1990s for products including nonstick cookware and food packaging and in industries such as fabric waterproofing, industrial manufacturing, and firefighting.
Legacy PFAS were phased out in the early 2000s, and novel PFAS were made to replace them. The term “novel” is independent of chemical properties and instead refers to when the chemicals’ production began, though novel PFAS typically have formulations meant to reduce their persistence in the environment. For example, many novel PFAS molecules have shorter chains of fluorinated carbons than their legacy counterparts.
Novel PFAS include possibly millions of different chemical structures, and their production and use are increasing globally.
In the United States and elsewhere, regulatory structures that limit PFAS production target specific chemicals, such that every new formulation by a company must be tested individually before restrictions are put in place. With companies continually conjuring new PFAS formulations—which environmental advocates often call “regrettable substitutions” for their sometimes harmful effects—understanding the fate and transport of novel PFAS is difficult and time-consuming. Research on the behavior of specific PFAS may be a drop in the bucket when millions of potential PFAS, with millions of potential behaviors, pose current and future risks to people and the environment.
Scientists like Sun are determined to untangle how the fate of these new chemicals differs from their predecessors. As Sun expected, the phaseout of legacy PFAS was reflected in the pilot whale tissue she tested. These results are good news; they clearly show that the bans on legacy PFAS are working.
“We’re still finding [older] compounds, but clearly, they are no longer as abundant in the environment as they used to be, which is a positive,” said Bridger Ruyle, an environmental engineer at New York University who studies PFAS and assisted Sun and her coauthors in deciding which methods to use for the new study.
But Sun and her colleagues also expected an overall increase in concentrations of novel PFAS—after all, production of these chemicals is higher than ever, and researchers finally had the analytical tools to catch them.
“The inference is, if it’s not in the whales, and it’s not in the ocean…where is it?”
That wasn’t what they found. Instead, all but two of the emerging PFAS they tested for were virtually nowhere to be seen in the whale tissue, leaving the scientists leading the study to wonder where novel PFAS were accumulating or if instrumentation was limiting their detection.
“We do know that the novel PFAS are being produced, which means they’re going somewhere. Where they are, and how exposed people and other wildlife are, is not as clear,” Sun said.
“The inference is, if it’s not in the whales, and it’s not in the ocean…where is it?” asked Elsie Sunderland, an environmental scientist at Harvard University and coauthor of the new study.
Sun and Sunderland’s question—asking where novel PFAS are going—is one scientists are probing from multiple angles. Those who study particle transport are asking how novel PFAS might travel through Earth’s water and air. Those on the chemistry side of the investigation are deducing how novel PFAS might break down. And those who monitor environments are looking for traces of novel PFAS in various corners of Earth.
The answers to their questions have direct, practical implications for human and environmental health and could indicate whether a growing proportion of harmful PFAS may be ending up in close proximity to humans—where we work and eat and breathe.
A Toxic Legacy
The chemical properties of PFAS have made the chemicals useful since the 1940s. These same properties also make them highly persistent—the most durable types may not break down in the environment for several thousand years.
PFAS are linked to certain cancers and other human health harms. Much of the available data linking PFAS to poor health come from analyses of legacy PFOA and PFOS. They show an association between increased exposure to these chemicals and altered immune and thyroid function, liver and kidney disease, reproductive system disruptions, and more.
Chemical manufacturers phased out production of legacy PFAS after scientific evidence emerged associating PFAS and human health harms, businesses began to lose money in massive lawsuits, and regulations tightened. Novel PFAS were intended to show properties similar to legacy PFAS but were meant to break down more easily in the environment (lower persistence) and accumulate less easily in living tissue (lower bioaccumulative ability), though studies have shown mixed results about whether novel PFAS are actually safer for humans or break down more easily.
Because PFAS production data are often proprietary, scientists who study PFAS, like Sun, must rely on partial inventories of PFAS production or reverse-engineer those numbers from observations in the environment.
“We call it chemical Whac-A-Mole.”
Without a clear list of the chemical structures of novel PFAS, scientists don’t always have the analytical standards necessary for routine detection. And once scientists do understand the behavior of a PFAS chemical, it may be quickly replaced by another, unknown alternative. “We call it chemical Whac-A-Mole,” Sunderland said.
Legacy PFAS tend to have a high affinity for water and typically end up in the ocean, the place scientists refer to as the chemicals’ “terminal sink.” Many legacy PFAS also entered the ocean through atmospheric transport such as rain or snow. But because of the sheer number of chemical formulas and the chemical differences between legacy and novel PFAS, the pathways that novel PFAS take through the environment are less clear.
Tracking the movement and accumulation of novel PFAS in the environment is crucial for understanding how these chemicals may affect ecological and human health.
Still, the science is inconclusive about whether novel PFAS are moving or accumulating differently than their legacy counterparts, whether they have a different terminal sink, and where that terminal sink may be.
Close to Home
One possible answer to the question of the missing novel PFAS may have to do with geography. The chemicals may not have reached pilot whales in the Faroes because something about the new chemistry has led them elsewhere in the environment. To Sun, evidence suggests “that a lot of these novel PFAS, which we know are being produced, may not be transporting out into this more remote environment either at all or as quickly.”
Novel PFAS might be accumulating closer to their sources—and closer to us. “It may simply be that some of the replacement PFAS don’t make it all the way out into the open ocean. But if they are still in the terrestrial environment and the near-coastal environment, then wildlife and people who live close to the sources can be exposed, said Frank Wania, an environmental chemist at the University of Toronto Scarborough.
For example, one study monitored PFAS in coastal beluga whales in Canada’s St. Lawrence Estuary, relatively close to human communities and PFAS manufacturing sources. The study showed increasing concentrations of unregulated novel PFAS in whale tissue from 2000 to 2017, while concentrations of legacy PFAS declined.
The suggestion that novel PFAS are accumulating close to human communities is supported by measurements of PFAS in human tissue, too. Studies show that a high proportion of detectable organofluorine chemicals in human tissue are increasingly unidentifiable, suggesting that some of the novel PFAS production “is in us,” Sunderland said.
Far and Away
Though there are some indications that novel PFAS may be retained closer to human communities, there are also reasons to think some novel PFAS chemistries have resulted in substances that can actually travel farther and more easily than their legacy counterparts.
Anna Kärrman, an environmental chemist at Örebro University in Sweden, said that some novel PFAS may be more easily transported in the environment: “The more novel chemistries are increasing the properties of being very mobile in water, very mobile in the atmosphere, and not necessarily very bioaccumulative.”
The mobility of novel PFAS was on full display in a 2020 study that Sunderland coauthored, in which researchers reported detecting hexafluoropropylene oxide-dimer acid, a novel PFAS chemical more commonly known as GenX, in the Arctic for the first time. GenX, produced by chemical manufacturer Chemours, was meant to replace the legacy compound PFOA. The 2020 study suggested GenX “has already moved quite a bit,” said Rainer Lohmann, a marine geochemist who leads the STEEP (Sources, Transport, Exposure and Effects of PFAS) Center at the University of Rhode Island.

The 2020 study also found higher concentrations of PFAS in the Arctic Ocean’s surface water, suggesting that the atmosphere was a particularly important transport pathway for chemical transport. This idea is supported by studies of High Arctic ice caps, which experience contamination only from atmospheric sources, and polar bear tissue. Atmospheric transport of novel PFAS is a subject “at the edge” of PFAS research, Sunderland said.
Wherever researchers look, they’re finding that atmospheric transport is an important pathway by which some PFAS, especially PFAS precursors—chemicals that break down in the environment and become PFAS (either novel or legacy)—move. One idea called the PAART (precursor atmospheric and reaction transport) theory was developed by Scott Mabury, an environmental chemist at the University of Toronto, and others. The PAART theory proposes that many of the harmful PFAS that end up in the most remote parts of Earth are the result of the breakdown of volatile precursor PFAS that have traveled in the atmosphere.
According to Lohmann, atmospheric transport means the ocean remains a terminal sink because many novel PFAS transported in rain or snow will ultimately be deposited in the ocean.
In this scenario, the question of why novel PFAS are not bioaccumulating in Faroese pilot whales remains a mystery. While Lohmann suggests the novel compounds simply don’t accumulate in living tissue, Sunderland isn’t sure that’s the whole story: “As apex predators, the whales are sentinels for what is available and being taken up from the ocean,” she wrote in an email. “Since we don’t see [novel PFAS], it seems unlikely there are large quantities of these chemicals present.”
Profuse PFAS
Another possible explanation for the surprising results of Sun’s whale study could be that there’s just a lag; that is, novel PFAS will end up in Faroe Island pilot whales someday but haven’t yet. Chemicals that could eventually end up in the ocean may be temporarily trapped in soils or recycled back into terrestrial ecosystems via sea spray aerosols, for example.
“The delay we are seeing in the ocean response may in fact be [PFAS] precursors being retained in source zones,” Sunderland wrote in an email. These chemicals may be “taking a really long time to be transformed into more mobile compounds.”
In their pilot whale study, Sun and her colleagues modeled the transport of PFAS to the subarctic and found a 10- to 20-year lag existed between the production of a legacy PFAS compound and its detection in whale tissue. We’re still within that range for many novel PFAS. Sun said she would have expected them to show up in pilot whale tissue by now if they behaved like their legacy counterparts, though it’s possible that it has taken time for the volume of novel PFAS production to ramp up, increasing the time it would take for the substances to be detected in tissues.

Still, the number of possible novel PFAS chemistries—again, there could be several million different compounds—makes it difficult to generalize how these new substances are, as a group, moving through the environment. “Because the exact structures of all [novel] PFAS remain unknown, some compounds may simply not be captured by the methods used,” Heidi Pickard, an environmental engineer at the consulting firm Ramboll and coauthor on the new whale study, wrote in an email.
Another reason novel PFAS are harder to study is that companies release lower concentrations of more kinds of the chemicals, rather than the “monstrously high” emissions of some legacy PFAS in the 1970s–1990s, noted Mabury, who was not involved in the new pilot whale study.
A New Regulatory Approach
According to Sun and Sunderland, cataloging differences between novel and legacy PFAS misses the broader point: We simply need to produce less PFAS. We’ve known for decades that PFAS harm human health, and some scientists have even argued that humans’ continual production and release of novel chemical compounds could drive Earth beyond a “safe operating space.”
“Researchers are critical for exposing the problem. But that, to me, is not the central issue here. The central issue here is a societal issue.”
Where scientists probe next may be less urgent than how policymakers decide to tackle the PFAS problem, Sunderland said: “Researchers are critical for exposing the problem. But that, to me, is not the central issue here. The central issue here is a societal issue.”
Chemical manufacturers are actively creating novel PFAS all the time. Kärrman, for example, has noticed patent applications for PFAS compounds with chemistries that “are nothing like we have seen before” that may start entering our environment in 5 or 10 years.
To Kärrman, that’s a reason for governments to push for chemical regulation based on properties such as persistence and bioaccumulation, rather than the chemical-by-chemical formula used in most countries, including the United States.
Such an approach has gotten traction in Europe via a proposal by the European Chemicals Agency to restrict the entire class of PFAS chemicals. The proposal is still under evaluation, and a final decision is expected by the end of the year.
In the United States, PFAS regulation and remediation are a key aspect of the Trump administration’s Make America Healthy Again movement, according to the EPA, and the federal government and some states already limit the concentrations of individual PFAS in drinking water. However, the EPA also said it planned to weaken some of those limits last year.
“We’re in a cycle of picking these regrettable alternatives [to legacy PFAS] and then figuring out that it was regrettable decades later,” Sunderland said. “We’re never going to catch up using this chemical-by-chemical approach.”
—Grace van Deelen (@gvd.bsky.social), Staff Writer
Citation: van Deelen, G. (2026), Chemical companies are churning out new PFAS. Where in the world are they ending up?, Eos, 107, https://doi.org/10.1029/2026EO260136. Published on 30 April 2026.
Text © 2026. The authors. CC BY-NC-ND 3.0
Except where otherwise noted, images are subject to copyright. Any reuse without express permission from the copyright owner is prohibited.


